DIAGNOSTIC MEDICAL MICROBIOLOGY
PRINCIPLES OF DIAGNOSTIC MEDICAL MICROBIOLOGY: INTRODUCTION
Diagnostic medical microbiology is concerned with the etiologic diagnosis of infection. Laboratory procedures used in the diagnosis of infectious disease in humans include the following:
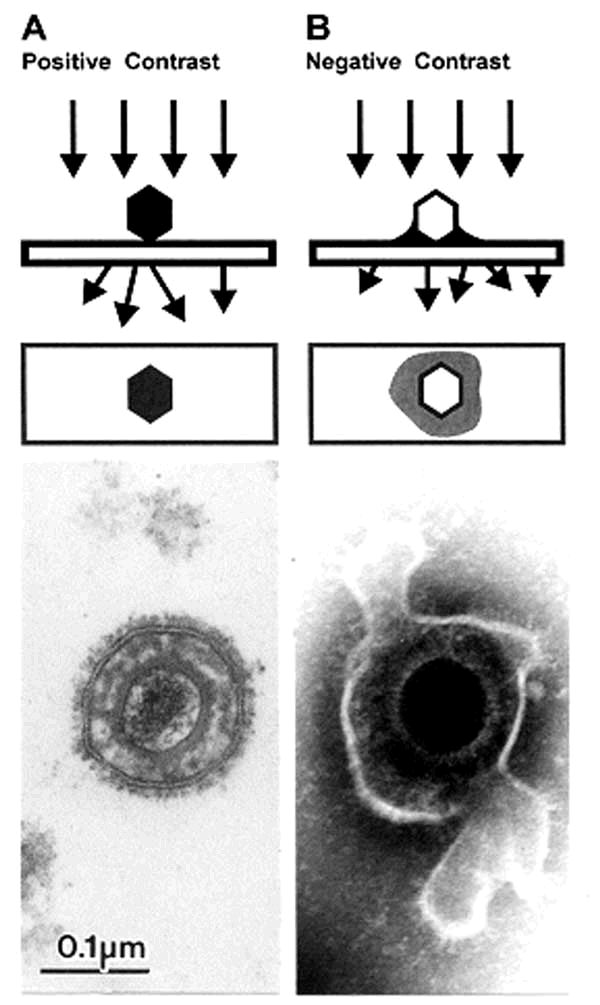
1. Morphologic identification of the agent in stains of specimens or sections of tissues (light and electron
microscopy).
2. Culture isolation and identification of the agent.
3. Detection of antigen from the agent by immunologic assay (latex agglutination, enzyme immunoassay
[EIA], etc) or by fluorescein-labeled (or peroxidase-labeled) antibody stains.
4. DNA -DNA or DNA -RNA hybridization to detect pathogen-specific genes in patients' specimens.
5. Detection and amplification of organism nucleic acid in patients' specimens.
6. Demonstration of meaningful antibody or cell-mediated immune responses to an infectious agent.
In the field of infectious diseases, laboratory test results depend largely on the quality of the specimen, the
timing and the care with which it is collected, and the technical proficiency and experience of laboratory
personnel. Although physicians should be competent to perform a few simple, crucial microbiologic tests—make and stain a smear , examine it microscopically , and streak a culture plate—technical details of the more involved procedures are usually left to the bacteriologist or virologist and the technicians on the staff . Physicians who deal with infectious processes must know when and how to take specimens, what laboratory examinations to request, and how to interpret the results.
COMMUNICATION BETWEEN PHYSICIAN & LABORATORY
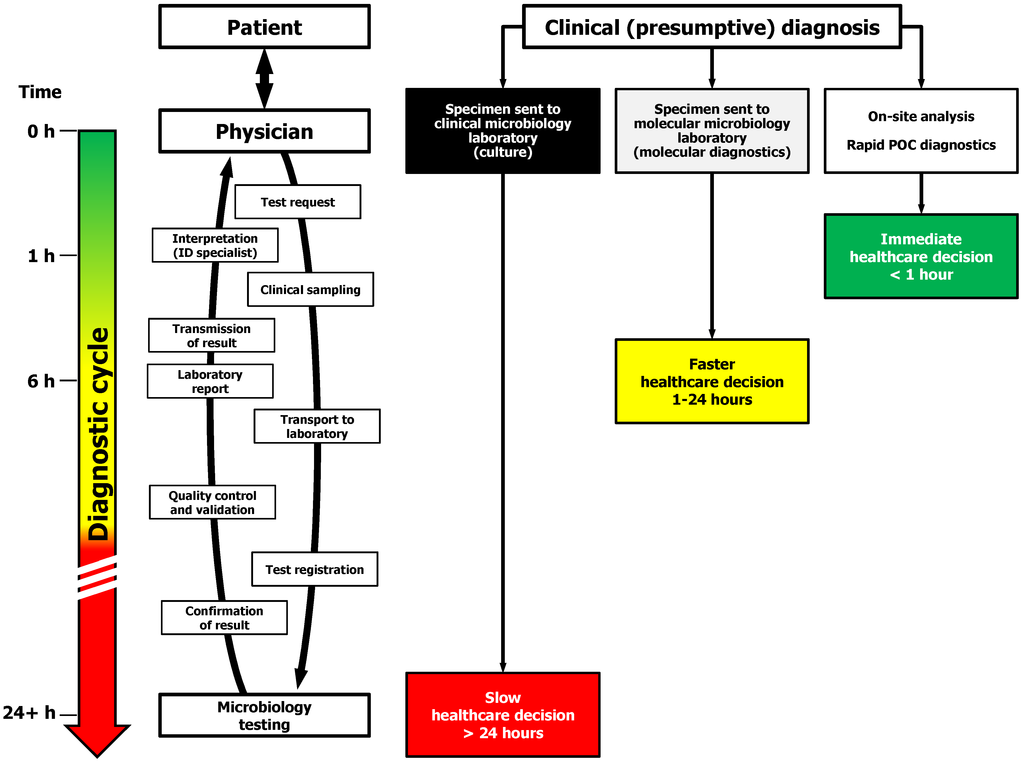
Diagnostic microbiology encompasses the characterization of thousands of agents that cause or are associated with infectious diseases. The techniques used to characterize infectious agents vary greatly depending upon the clinical syndrome and the type of agent being considered, be it virus, bacterium, fungus, or other parasite. Because no single test will permit isolation or characterization of all potential pathogens, clinical information is much more important for diagnostic microbiology than it is for clinical chemistry or hematology . The clinician must make a tentative diagnosis rather than wait until laboratory results are available. When tests are requested, the physician should inform the laboratory staff of the tentative diagnosis (type of infection or infectious agent suspected). Proper labeling of specimens includes such clinical data as well as the patient's identifying data (at least two methods of definitive identification) and the requesting physician's name and pertinent contact information. Many pathogenic microorganisms grow slowly , and days or even weeks may elapse before they are isolated and identified. Treatment cannot be deferred until this process is complete. After obtaining the proper specimens and informing the laboratory of the tentative clinical diagnosis, the physician should begin treatment with drugs aimed at the organism thought to be responsible for the patient's illness.As the laboratory staff begins to obtain results, they inform the physician, who can then reevaluate the diagnosis and clinical course of the patient and perhaps make changes in the therapeutic program. This "feedback" information from the laboratory consists of preliminary reports of the results of individual steps in the isolation and identification of the causative agent.
DIAGNOSIS OF BACTERIAL & FUNGAL INFECTIONS
Specimens
Laboratory examination usually includes microscopic study of fresh unstained and stained materials and
preparation of cultures with conditions suitable for growth of a wide variety of microorganisms, including the
type of organism most likely to be causative based on clinical evidence. If a microorganism is isolated,
complete identification may then be pursued. Isolated microorganisms may be tested for susceptibility to
antimicrobial drugs. When significant pathogens are isolated before treatment, follow-up laboratory
examinations during and after treatment may be appropriate.
A properly collected specimen is the single most important step in the diagnosis of an infection, because the
results of diagnostic tests for infectious diseases depend upon the selection, timing, and method of collection of specimens. Bacteria and fungi grow and die, are susceptible to many chemicals, and can be found at different anatomic sites and in different body fluids and tissues during the course of infectious diseases. Because isolation of the agent is so important in the formulation of a diagnosis, the specimen must be obtained from the site most likely to yield the agent at that particular stage of illness and must be handled in such a way as to favor the agent's survival and growth. F or each type of specimen, suggestions for optimal handling are given in the following paragraphs and in the section on diagnosis by anatomic site, below. Recovery of bacteria and fungi is most significant if the agent is isolated from a site normally devoid of microorganisms (a normally sterile area). Any type of microorganism cultured from blood, cerebrospinal fluid, joint fluid, or the pleural cavity is a significant diagnostic finding. Conversely , many parts of the body have normal microbiota that may be altered by endogenous or exogenous influences. Recovery of potential pathogens from the respiratory , gastrointestinal, or genitourinary tracts; from wounds; or from the skin must be considered in the context of the normal microbiota of each particular site. Microbiologic data must be correlated with clinical information in order to arrive at a meaningful interpretation of results.
A few general rules apply to all specimens:
1. The quantity of material must be adequate.
2. The sample should be representative of the infectious process (eg, sputum, not saliva; pus from the
underlying lesion, not from its sinus tract; a swab from the depth of the wound, not from its surface).
3. Contamination of the specimen must be avoided by using only sterile equipment and aseptic precautions.
4. The specimen must be taken to the laboratory and examined promptly . Special transport media may be
helpful.
5. Meaningful specimens to diagnose bacterial and fungal infections must be secured before antimicrobial
drugs are administered. If antimicrobial drugs are given before specimens are taken for microbiologic
study drug therapy may have to be stopped and repeat specimens obtained several days later .
The type of specimen to be examined is determined by the presenting clinical picture. If symptoms or signs
point to involvement of one organ system, specimens are obtained from that source. In the absence of
localizing signs or symptoms, repeated blood samples for culturing are taken first, and specimens from other
sites are then considered in sequence, depending in part upon the likelihood of involvement of a given organ
system in a given patient and in part upon the ease of obtaining specimens.
Microscopy & Stains
Microscopic examination of stained or unstained specimens is a relatively simple and inexpensive but much less sensitive method than culture for detection of small numbers of bacteria. A specimen must contain at least 10^5 organisms per milliliter before it is likely that organisms will be seen on a smear . Liquid medium containing 10^5 organisms per milliliter does not appear turbid to the eye. Specimens containing 10^2 to 10^3 organisms per milliliter produce growth on solid media, and those containing ten or fewer bacteria per milliliter may produce growth in liquid media. Gram staining is a very useful procedure in diagnostic microbiology . Most specimens submitted when bacterial infection is suspected should be smeared on glass slides, Gram-stained, and examined microscopically. On microscopic examination, the Gram reaction (purple-blue indicates gram-positive organisms; red, gram-negative) and morphology (shape: cocci,
rods, fusiform, or other of bacteria should be noted. The appearance of bacteria on Gram stained smears does not permit identification of species. Reports of gram-positive cocci in chains are suggestive
of , but not definitive for , streptococcal species; gram-positive cocci in clusters suggest a staphylococcal species. Gram-negative rods can be large, small, or even coccobacillary . Some nonviable gram-positive bacteria can stain gram-negatively. Typically , bacterial morphology has been defined using organisms grown on agar. However , bacteria in body fluids or tissue can have highly variable morphology.
Gram & Acid-Fast Staining Methods
Gram stain
(1) Fix smear by heat or using methanol.
(2) Cover with crystal violet.
(3) Wash with water . Do not blot.
(4) Cover with Gram's iodine.
(5) Wash with water . Do not blot.
(6) Decolorize for 10–30 seconds with gentle agitation in acetone (30 mL) and alcohol (70 mL).
(7) Wash with water . Do not blot.
(8) Cover for 10–30 seconds with safranin (2.5% solution in 95% alcohol).
(9) Wash with water and let dry . Ziehl-Neelsen acid-fast stain
(1) Fix smear by heat.
(2) Cover with carbol fuchsin, steam gently for 5 minutes over direct flame (or for 20 minutes over a water
bath). Do not permit slides to boil or dry out.
(3) Wash with deionized water .
(4) Decolorize in 3.0% acid-alcohol (95% ethanol and 3.0% hydrochloric acid) until only a faint pink color
remains.
(5) Wash with water .
(6) Counterstain for 1 minute with Loeffler's methylene blue.
(7) Wash with deionized water and let dry .
Kinyoun carbolfuchsin acid-fast stain
(1) Formula: 4 g basic fuchsin, 8 g phenol, 20 mL 95% alcohol, 100 mL distilled water .
(2) Stain fixed smear for 3 minutes (no heat necessary) and continue as with Ziehl-Neelsen stain.
Specimens submitted for examination for mycobacteria should be stained for acid-fast organisms, using either Ziehl-Neelsen stain or Kinyoun stain. An alternative fluorescent stain for mycobacteria,
auramine-rhodamine stain, is more sensitive than other stains for acid-fast organisms but requires fluorescence microscopy and, if results are positive, confirmation of morphology with an acid-fast stain.
Immunofluorescent antibody (IF) staining is useful in the identification of many microorganisms. Such
procedures are more specific than other staining techniques but also more cumbersome to perform. The
fluorescein-labeled antibodies in common use are made from antisera produced by injecting animals with whole organisms or complex antigen mixtures. The resultant polyclonal antibodies may react with multiple
antigens on the organism that was injected and may also cross-react with antigens of other microorganisms or possibly with human cells in the specimen. Quality control is important to minimize nonspecific IF staining. Use of monoclonal antibodies may circumvent the problem of nonspecific staining. IF staining is most useful in confirming the presence of specific organisms such as Bordetella pertussis or Legionella pneumophila in colonies isolated on culture media. The use of direct IF staining on specimens from patients is more difficult and less specific.
Cited By Anil Bhujel.
Bsc Microbiology, TU.
Microbiology Student At PBPC, Nayabazzar-9, Pokhara.
SOME SUGGESTED REFERENCES:
www.ncbi.nlm.nih.gov › NCBI › Literature › Bookshelf
https://sites.google.com/.../allmicrobiologysite/diagnostic-medical-microb...
www.slideshare.net/chhaya30/diagnostic-microbiology
biomedicgen.files.wordpress.com/.../diagnostic-medical-microbiology-cli...
accessmedicine.mhmedical.com/content.aspx?bookid=504§ionid...
en.wikipedia.org/wiki/Medical_microbiology
cc.shsmu.edu.cn/G2S/Template/ShowNoMeau.aspx?type=3...pdf...
tainguyenso.vnu.edu.vn/xmlui/bitstream/handle/.../01071000101.pdf?...
en.wikipedia.org/wiki/Staining
https://www.microscopeworld.com/t-microscope_slide_staining.aspx
www.microscopemaster.com/cell-staining-microscopy.html
Diagnostic medical microbiology is concerned with the etiologic diagnosis of infection. Laboratory procedures used in the diagnosis of infectious disease in humans include the following:
1. Morphologic identification of the agent in stains of specimens or sections of tissues (light and electron
microscopy).
2. Culture isolation and identification of the agent.
3. Detection of antigen from the agent by immunologic assay (latex agglutination, enzyme immunoassay
[EIA], etc) or by fluorescein-labeled (or peroxidase-labeled) antibody stains.
4. DNA -DNA or DNA -RNA hybridization to detect pathogen-specific genes in patients' specimens.
5. Detection and amplification of organism nucleic acid in patients' specimens.
6. Demonstration of meaningful antibody or cell-mediated immune responses to an infectious agent.
In the field of infectious diseases, laboratory test results depend largely on the quality of the specimen, the
timing and the care with which it is collected, and the technical proficiency and experience of laboratory
personnel. Although physicians should be competent to perform a few simple, crucial microbiologic tests—make and stain a smear , examine it microscopically , and streak a culture plate—technical details of the more involved procedures are usually left to the bacteriologist or virologist and the technicians on the staff . Physicians who deal with infectious processes must know when and how to take specimens, what laboratory examinations to request, and how to interpret the results.
COMMUNICATION BETWEEN PHYSICIAN & LABORATORY
Diagnostic microbiology encompasses the characterization of thousands of agents that cause or are associated with infectious diseases. The techniques used to characterize infectious agents vary greatly depending upon the clinical syndrome and the type of agent being considered, be it virus, bacterium, fungus, or other parasite. Because no single test will permit isolation or characterization of all potential pathogens, clinical information is much more important for diagnostic microbiology than it is for clinical chemistry or hematology . The clinician must make a tentative diagnosis rather than wait until laboratory results are available. When tests are requested, the physician should inform the laboratory staff of the tentative diagnosis (type of infection or infectious agent suspected). Proper labeling of specimens includes such clinical data as well as the patient's identifying data (at least two methods of definitive identification) and the requesting physician's name and pertinent contact information. Many pathogenic microorganisms grow slowly , and days or even weeks may elapse before they are isolated and identified. Treatment cannot be deferred until this process is complete. After obtaining the proper specimens and informing the laboratory of the tentative clinical diagnosis, the physician should begin treatment with drugs aimed at the organism thought to be responsible for the patient's illness.As the laboratory staff begins to obtain results, they inform the physician, who can then reevaluate the diagnosis and clinical course of the patient and perhaps make changes in the therapeutic program. This "feedback" information from the laboratory consists of preliminary reports of the results of individual steps in the isolation and identification of the causative agent.
DIAGNOSIS OF BACTERIAL & FUNGAL INFECTIONS
Specimens
Laboratory examination usually includes microscopic study of fresh unstained and stained materials and
preparation of cultures with conditions suitable for growth of a wide variety of microorganisms, including the
type of organism most likely to be causative based on clinical evidence. If a microorganism is isolated,
complete identification may then be pursued. Isolated microorganisms may be tested for susceptibility to
antimicrobial drugs. When significant pathogens are isolated before treatment, follow-up laboratory
examinations during and after treatment may be appropriate.
A properly collected specimen is the single most important step in the diagnosis of an infection, because the
results of diagnostic tests for infectious diseases depend upon the selection, timing, and method of collection of specimens. Bacteria and fungi grow and die, are susceptible to many chemicals, and can be found at different anatomic sites and in different body fluids and tissues during the course of infectious diseases. Because isolation of the agent is so important in the formulation of a diagnosis, the specimen must be obtained from the site most likely to yield the agent at that particular stage of illness and must be handled in such a way as to favor the agent's survival and growth. F or each type of specimen, suggestions for optimal handling are given in the following paragraphs and in the section on diagnosis by anatomic site, below. Recovery of bacteria and fungi is most significant if the agent is isolated from a site normally devoid of microorganisms (a normally sterile area). Any type of microorganism cultured from blood, cerebrospinal fluid, joint fluid, or the pleural cavity is a significant diagnostic finding. Conversely , many parts of the body have normal microbiota that may be altered by endogenous or exogenous influences. Recovery of potential pathogens from the respiratory , gastrointestinal, or genitourinary tracts; from wounds; or from the skin must be considered in the context of the normal microbiota of each particular site. Microbiologic data must be correlated with clinical information in order to arrive at a meaningful interpretation of results.
A few general rules apply to all specimens:
1. The quantity of material must be adequate.
2. The sample should be representative of the infectious process (eg, sputum, not saliva; pus from the
underlying lesion, not from its sinus tract; a swab from the depth of the wound, not from its surface).
3. Contamination of the specimen must be avoided by using only sterile equipment and aseptic precautions.
4. The specimen must be taken to the laboratory and examined promptly . Special transport media may be
helpful.
5. Meaningful specimens to diagnose bacterial and fungal infections must be secured before antimicrobial
drugs are administered. If antimicrobial drugs are given before specimens are taken for microbiologic
study drug therapy may have to be stopped and repeat specimens obtained several days later .
The type of specimen to be examined is determined by the presenting clinical picture. If symptoms or signs
point to involvement of one organ system, specimens are obtained from that source. In the absence of
localizing signs or symptoms, repeated blood samples for culturing are taken first, and specimens from other
sites are then considered in sequence, depending in part upon the likelihood of involvement of a given organ
system in a given patient and in part upon the ease of obtaining specimens.
Microscopy & Stains
Microscopic examination of stained or unstained specimens is a relatively simple and inexpensive but much less sensitive method than culture for detection of small numbers of bacteria. A specimen must contain at least 10^5 organisms per milliliter before it is likely that organisms will be seen on a smear . Liquid medium containing 10^5 organisms per milliliter does not appear turbid to the eye. Specimens containing 10^2 to 10^3 organisms per milliliter produce growth on solid media, and those containing ten or fewer bacteria per milliliter may produce growth in liquid media. Gram staining is a very useful procedure in diagnostic microbiology . Most specimens submitted when bacterial infection is suspected should be smeared on glass slides, Gram-stained, and examined microscopically. On microscopic examination, the Gram reaction (purple-blue indicates gram-positive organisms; red, gram-negative) and morphology (shape: cocci,
rods, fusiform, or other of bacteria should be noted. The appearance of bacteria on Gram stained smears does not permit identification of species. Reports of gram-positive cocci in chains are suggestive
of , but not definitive for , streptococcal species; gram-positive cocci in clusters suggest a staphylococcal species. Gram-negative rods can be large, small, or even coccobacillary . Some nonviable gram-positive bacteria can stain gram-negatively. Typically , bacterial morphology has been defined using organisms grown on agar. However , bacteria in body fluids or tissue can have highly variable morphology.
Gram & Acid-Fast Staining Methods
Gram stain
(1) Fix smear by heat or using methanol.
(2) Cover with crystal violet.
(3) Wash with water . Do not blot.
(4) Cover with Gram's iodine.
(5) Wash with water . Do not blot.
(6) Decolorize for 10–30 seconds with gentle agitation in acetone (30 mL) and alcohol (70 mL).
(7) Wash with water . Do not blot.
(8) Cover for 10–30 seconds with safranin (2.5% solution in 95% alcohol).
(9) Wash with water and let dry . Ziehl-Neelsen acid-fast stain
(1) Fix smear by heat.
(2) Cover with carbol fuchsin, steam gently for 5 minutes over direct flame (or for 20 minutes over a water
bath). Do not permit slides to boil or dry out.
(3) Wash with deionized water .
(4) Decolorize in 3.0% acid-alcohol (95% ethanol and 3.0% hydrochloric acid) until only a faint pink color
remains.
(5) Wash with water .
(6) Counterstain for 1 minute with Loeffler's methylene blue.
(7) Wash with deionized water and let dry .
Kinyoun carbolfuchsin acid-fast stain
(1) Formula: 4 g basic fuchsin, 8 g phenol, 20 mL 95% alcohol, 100 mL distilled water .
(2) Stain fixed smear for 3 minutes (no heat necessary) and continue as with Ziehl-Neelsen stain.
Specimens submitted for examination for mycobacteria should be stained for acid-fast organisms, using either Ziehl-Neelsen stain or Kinyoun stain. An alternative fluorescent stain for mycobacteria,
auramine-rhodamine stain, is more sensitive than other stains for acid-fast organisms but requires fluorescence microscopy and, if results are positive, confirmation of morphology with an acid-fast stain.
Immunofluorescent antibody (IF) staining is useful in the identification of many microorganisms. Such
procedures are more specific than other staining techniques but also more cumbersome to perform. The
fluorescein-labeled antibodies in common use are made from antisera produced by injecting animals with whole organisms or complex antigen mixtures. The resultant polyclonal antibodies may react with multiple
antigens on the organism that was injected and may also cross-react with antigens of other microorganisms or possibly with human cells in the specimen. Quality control is important to minimize nonspecific IF staining. Use of monoclonal antibodies may circumvent the problem of nonspecific staining. IF staining is most useful in confirming the presence of specific organisms such as Bordetella pertussis or Legionella pneumophila in colonies isolated on culture media. The use of direct IF staining on specimens from patients is more difficult and less specific.
Cited By Anil Bhujel.
Bsc Microbiology, TU.
Microbiology Student At PBPC, Nayabazzar-9, Pokhara.
SOME SUGGESTED REFERENCES:
www.ncbi.nlm.nih.gov › NCBI › Literature › Bookshelf
https://sites.google.com/.../allmicrobiologysite/diagnostic-medical-microb...
www.slideshare.net/chhaya30/diagnostic-microbiology
biomedicgen.files.wordpress.com/.../diagnostic-medical-microbiology-cli...
accessmedicine.mhmedical.com/content.aspx?bookid=504§ionid...
en.wikipedia.org/wiki/Medical_microbiology
cc.shsmu.edu.cn/G2S/Template/ShowNoMeau.aspx?type=3...pdf...
tainguyenso.vnu.edu.vn/xmlui/bitstream/handle/.../01071000101.pdf?...
en.wikipedia.org/wiki/Staining
https://www.microscopeworld.com/t-microscope_slide_staining.aspx
www.microscopemaster.com/cell-staining-microscopy.html





Comments
For more useful information about Biotechnology visit BioTechBug
For making bright career in Biotechnology you should need Biotechnology Scholarships .
Biotechnology Courses in India