CONTROL OF MICROBIAL INFECTIONS
ANTIVIRAL DRUG RESISTANCE:
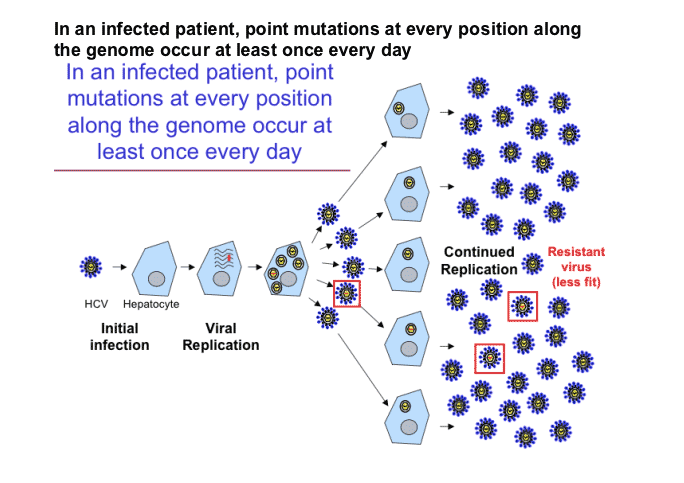
When an antibacterial or antiviral agent acts at a single target site then resistance can develop through
mutations at that site. The high mutation rates in RNA viruses will mean that such problems will occur
more readily than with DNA-based bacteria and this is witnessed in HIV therapies. Again, as with
antibiotic policies, certain manoeuvres can be employed to minimise resistance developing. These
include the use of combinations of antiviral compounds, switching between two unrelated agents
(although sequential use of antivirals has been criticised for simply promoting resistance rather than
preventing it) and the use of high concentrations to prevent any viral replication (‘knock out’). This,
theoretically, prevents any viable mutants being released.
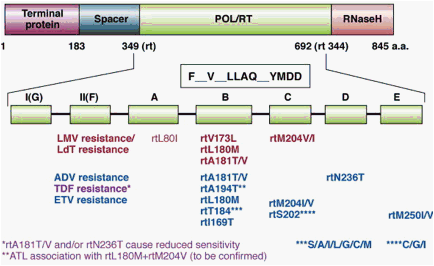
Resistance to antiviral agents will result from mutations in the viral genome. The modified proteins may
act on cell events that occur upstream of the active compound (e.g. mutations in viral thymidine kinase
will reduce its affinity of binding to the nucleoside) or mutations may occur in the target protein itself (e.
g. retroviral reverse transcriptase). Whilst mutation is a frequent event in RNA viruses, and resistance
develops frequently during the course of an infection, the spontaneous mutation events in a DNA virus
like Herpes simplex will be of much lower frequency. Note that latent Herpes simplex virus does not
express TK or DNA polymerase, hence the virus remains unaffected by the drug.
The laboratory testing for resistance to antiviral agents has a number of problems. Direct testing of the
inhibitory concentrations of antiviral agent (phenotypic testing) gives a concentration that is active, at
least under laboratory conditions. Phenotypic testing requires that the organism has been cultured and
this is not possible for those viruses that have not yet been successfully cultured in vitro. Molecular
mechanisms avoid this problem. Genotype testing can, in theory, be carried out on clinical samples as
well as cultured virus. Polymerase chain detection for resistant gene sequences are increasingly available
and are less technically demanding than phenotypic tests. The interpretation of both types of testing is
somewhat arbitrary and, like antibiotic susceptibility testing in bacteria, will only indicate possible outcomes or provide frames of reference.
SURVEILLANCE:
In the latter half of the 1980s, the incidence of tuberculosis had increased to ‘epidemic’ proportions in a
developed city, New York, in the most prosperous country in the world. How could such a well recognised infectious disease against which an effective vaccine and a range of antibiotics are available
spread unnoticed in such a city? The reasons illustrate how good public health infrastructure can be
undermined if unsupported. Famously associated with poverty, tuberculosis had steadily increased in a
place where HIV was endemic amongst people living in overcrowded, poor housing. The treatment of
tuberculosis uses three antibiotics over 6 months and patients with pulmonary disease who are the infectious sources quickly become non-infectious following antibiotic treatment. If patients fail to complete the treatment, they will relapse into infectious cases and
antibiotic resistance may result. Of the measures taken, DOT (direct observation of therapy) was
crucial to the management of the epidemic. DOT targets the source by effecting a reduction of the
proportion of infectious cases. One of the reasons for the return of tuberculosis was the reduced funding
to public health departments responsible for monitoring (surveillance) and treating the disease. Partly
this was a consequence of years of success in reducing the incidence of tuberculosis such that
complacency had set in. Not surprisingly the cost of bringing the epidemic under control was more than
the savings made in the first place.
The value of quarantine measures to screen imported animals for infectious diseases needs
consideration. The ancient practice of retaining ships in port for 40 days (‘quarantina’)
applied to all trade, not just to animals, and was driven mostly by a need to prevent the spread
of plague. With the spatial isolation of an island, quarantine can prevent the introduction of a
disease. With increasing international travel by air, the principle has been undermined. The
building of a tunnel under the English channel to connect the United Kingdom with the rest of
Europe means the isolation is breached again.
FUTURE EPIDEMICS
Epidemics have been a regular feature of human existence throughout history, so it is easy to state with
confidence that they will continue to be so. The reasons for such a gloomy outlook are not difficult to find. As populations have increased in size, particularly in cities, so has the incidence of
infectious disease. The proportion of people infected by certain infectious diseases (tuberculosis is again
an example) may fall, but the absolute numbers of people infected will still increase. The present
population forecasts indicate increasing numbers of large (greater than 10 million people) cities, mostly
in ports and concentrated in developing countries. Other than providing a critical population threshold to
sustain diseases like measles, sexually transmitted diseases and the common cold, large cities need
global commerce to support them, as well as attracting migration, which both increase the inward and
outward flux of people, goods and microbes. The public health infrastructure needs to be appropriately
sized and funded to prevent the resurgence of old infections that, although once under control, will
readily return without continued control. The expansion of the areas of known infectious disease is a
result of expanding urban slums into the country such that the associated risks of deforestation and
monoculture of crops all result in disturbing zoonotic cycles. It is also depressing to note the regular
appearance of epidemics that accompany war, a habit that, far from diminishing, seems to be on the
increase and risks employing biological weapons. Although poverty is famously linked with poor health,
notably tuberculosis, wealthy nations are not excluded from risk as advances in medicine keep more people alive with increasing opportunities for infections.
EXAMPLES OF CHANGING PATTERNS IN INFECTIOUS DISEASE
• Previously unrecognised diseases: Lyme disease (Borrelia burgdoferi), Helicobacter
pylori gastritis.
• Increasing zone of disease: increasing malaria and Dengue due to the spread of
mosquito vectors into new areas.
• Resurgence of ‘old’ infections: epidemic wave of diphtheria in Europe affecting the
old communist bloc.
• Apparent synergy in tuberculosis with HIV in AIDS patients.
• Increasing incidence of drug-resistant tuberculosis.
• Food-borne infections increasing with globalisation of food products.
International surveillance will become increasingly important as the world shrinks through globalisation
in order that infectious diseases can be tracked. The bioterrorist threat, exemplified by the crippling cost of sending a few samples of anthrax spores through the post, is a further call to adequate microbiological infrastructure and expertise. The factors that are important in protecting the public from biological weapons are similar to those that are considered in all microbiology laboratories. The need for microbiologists is obvious but should that idea need reinforcement then it is worth noting
that the cost of eradication of poliomyelitis in the USA has paid for itself every 26 days since 1977. The
financial losses to a country incurred through infectious disease every year may yet make governments
take notice.
Having described the human activities that promote infectious disease, what are the features of the
organisms that will continue to create problems? RNA viruses are the organisms with the highest
mutation rates. It can be expected that such organisms will continue to cause epidemics, just as HIV has
been the most frightening of recent times because of the genetic variation that occurs through inaccurate
transcription and genetic reassortment. HIV has demonstrated the potential impact that an RNA virus
can have if it is able to cross the species barrier (from apes to humans). The risk of mutational events
resulting in greater virulence in bacteria and fungi is less than for RNA viruses, but the increasing
recognition that toxins and pathogenicity islands are coded for on mobile genetic elements means that
bacteria will continue to present threats. The increase in antibiotic resistance genes through horizontal
gene transfer over the last 50 years reminds us that we have not yet overcome the existing microbial
threats. HAZARD GROUPS
In order to protect the staff in microbiology laboratories as well as the public and the environment,
micro-organisms have been categorised into four Hazard Groups. The groups, in increasing order of
hazard, are as follows:
Group 1: Unlikely to cause human disease.
Group 2: Can cause disease and may be a hazard to employees.
Unlikely to spread to the community.
Effective prophylaxis or treatment available.
Group 3: Can cause human disease.
Risk of spread to the community.
Effective prophylaxis or treatment available.
Group 4: Can cause severe human disease.
Likely risk of spread into the community.
No effective prophylaxis and treatment.
The micro-organisms considered the most effective biological weapons are chosen because
they:
• are easily disseminated into the public or are highly infectious (readily transmitted
from person to person).
• cause high mortality.
• require special action for public health control
The organisms mostly fall into Hazard Group 4. Viruses such as Variola major virus
(smallpox) and Ebola and Marburg viruses are not easily treated with antiviral agents but
there are effective vaccines available (although not routinely used). Bacillus anthracis and Yersinia pestis(the aetiological agent of plague) can be treated with antibiotics.
.
When an antibacterial or antiviral agent acts at a single target site then resistance can develop through
mutations at that site. The high mutation rates in RNA viruses will mean that such problems will occur
more readily than with DNA-based bacteria and this is witnessed in HIV therapies. Again, as with
antibiotic policies, certain manoeuvres can be employed to minimise resistance developing. These
include the use of combinations of antiviral compounds, switching between two unrelated agents
(although sequential use of antivirals has been criticised for simply promoting resistance rather than
preventing it) and the use of high concentrations to prevent any viral replication (‘knock out’). This,
theoretically, prevents any viable mutants being released.
Resistance to antiviral agents will result from mutations in the viral genome. The modified proteins may
act on cell events that occur upstream of the active compound (e.g. mutations in viral thymidine kinase
will reduce its affinity of binding to the nucleoside) or mutations may occur in the target protein itself (e.
g. retroviral reverse transcriptase). Whilst mutation is a frequent event in RNA viruses, and resistance
develops frequently during the course of an infection, the spontaneous mutation events in a DNA virus
like Herpes simplex will be of much lower frequency. Note that latent Herpes simplex virus does not
express TK or DNA polymerase, hence the virus remains unaffected by the drug.
The laboratory testing for resistance to antiviral agents has a number of problems. Direct testing of the
inhibitory concentrations of antiviral agent (phenotypic testing) gives a concentration that is active, at
least under laboratory conditions. Phenotypic testing requires that the organism has been cultured and
this is not possible for those viruses that have not yet been successfully cultured in vitro. Molecular
mechanisms avoid this problem. Genotype testing can, in theory, be carried out on clinical samples as
well as cultured virus. Polymerase chain detection for resistant gene sequences are increasingly available
and are less technically demanding than phenotypic tests. The interpretation of both types of testing is
somewhat arbitrary and, like antibiotic susceptibility testing in bacteria, will only indicate possible outcomes or provide frames of reference.
SURVEILLANCE:
In the latter half of the 1980s, the incidence of tuberculosis had increased to ‘epidemic’ proportions in a
developed city, New York, in the most prosperous country in the world. How could such a well recognised infectious disease against which an effective vaccine and a range of antibiotics are available
spread unnoticed in such a city? The reasons illustrate how good public health infrastructure can be
undermined if unsupported. Famously associated with poverty, tuberculosis had steadily increased in a
place where HIV was endemic amongst people living in overcrowded, poor housing. The treatment of
tuberculosis uses three antibiotics over 6 months and patients with pulmonary disease who are the infectious sources quickly become non-infectious following antibiotic treatment. If patients fail to complete the treatment, they will relapse into infectious cases and
antibiotic resistance may result. Of the measures taken, DOT (direct observation of therapy) was
crucial to the management of the epidemic. DOT targets the source by effecting a reduction of the
proportion of infectious cases. One of the reasons for the return of tuberculosis was the reduced funding
to public health departments responsible for monitoring (surveillance) and treating the disease. Partly
this was a consequence of years of success in reducing the incidence of tuberculosis such that
complacency had set in. Not surprisingly the cost of bringing the epidemic under control was more than
the savings made in the first place.
The value of quarantine measures to screen imported animals for infectious diseases needs
consideration. The ancient practice of retaining ships in port for 40 days (‘quarantina’)
applied to all trade, not just to animals, and was driven mostly by a need to prevent the spread
of plague. With the spatial isolation of an island, quarantine can prevent the introduction of a
disease. With increasing international travel by air, the principle has been undermined. The
building of a tunnel under the English channel to connect the United Kingdom with the rest of
Europe means the isolation is breached again.
FUTURE EPIDEMICS
Epidemics have been a regular feature of human existence throughout history, so it is easy to state with
confidence that they will continue to be so. The reasons for such a gloomy outlook are not difficult to find. As populations have increased in size, particularly in cities, so has the incidence of
infectious disease. The proportion of people infected by certain infectious diseases (tuberculosis is again
an example) may fall, but the absolute numbers of people infected will still increase. The present
population forecasts indicate increasing numbers of large (greater than 10 million people) cities, mostly
in ports and concentrated in developing countries. Other than providing a critical population threshold to
sustain diseases like measles, sexually transmitted diseases and the common cold, large cities need
global commerce to support them, as well as attracting migration, which both increase the inward and
outward flux of people, goods and microbes. The public health infrastructure needs to be appropriately
sized and funded to prevent the resurgence of old infections that, although once under control, will
readily return without continued control. The expansion of the areas of known infectious disease is a
result of expanding urban slums into the country such that the associated risks of deforestation and
monoculture of crops all result in disturbing zoonotic cycles. It is also depressing to note the regular
appearance of epidemics that accompany war, a habit that, far from diminishing, seems to be on the
increase and risks employing biological weapons. Although poverty is famously linked with poor health,
notably tuberculosis, wealthy nations are not excluded from risk as advances in medicine keep more people alive with increasing opportunities for infections.
EXAMPLES OF CHANGING PATTERNS IN INFECTIOUS DISEASE
• Previously unrecognised diseases: Lyme disease (Borrelia burgdoferi), Helicobacter
pylori gastritis.
• Increasing zone of disease: increasing malaria and Dengue due to the spread of
mosquito vectors into new areas.
• Resurgence of ‘old’ infections: epidemic wave of diphtheria in Europe affecting the
old communist bloc.
• Apparent synergy in tuberculosis with HIV in AIDS patients.
• Increasing incidence of drug-resistant tuberculosis.
• Food-borne infections increasing with globalisation of food products.
International surveillance will become increasingly important as the world shrinks through globalisation
in order that infectious diseases can be tracked. The bioterrorist threat, exemplified by the crippling cost of sending a few samples of anthrax spores through the post, is a further call to adequate microbiological infrastructure and expertise. The factors that are important in protecting the public from biological weapons are similar to those that are considered in all microbiology laboratories. The need for microbiologists is obvious but should that idea need reinforcement then it is worth noting
that the cost of eradication of poliomyelitis in the USA has paid for itself every 26 days since 1977. The
financial losses to a country incurred through infectious disease every year may yet make governments
take notice.
Having described the human activities that promote infectious disease, what are the features of the
organisms that will continue to create problems? RNA viruses are the organisms with the highest
mutation rates. It can be expected that such organisms will continue to cause epidemics, just as HIV has
been the most frightening of recent times because of the genetic variation that occurs through inaccurate
transcription and genetic reassortment. HIV has demonstrated the potential impact that an RNA virus
can have if it is able to cross the species barrier (from apes to humans). The risk of mutational events
resulting in greater virulence in bacteria and fungi is less than for RNA viruses, but the increasing
recognition that toxins and pathogenicity islands are coded for on mobile genetic elements means that
bacteria will continue to present threats. The increase in antibiotic resistance genes through horizontal
gene transfer over the last 50 years reminds us that we have not yet overcome the existing microbial
threats. HAZARD GROUPS
In order to protect the staff in microbiology laboratories as well as the public and the environment,
micro-organisms have been categorised into four Hazard Groups. The groups, in increasing order of
hazard, are as follows:
Group 1: Unlikely to cause human disease.
Group 2: Can cause disease and may be a hazard to employees.
Unlikely to spread to the community.
Effective prophylaxis or treatment available.
Group 3: Can cause human disease.
Risk of spread to the community.
Effective prophylaxis or treatment available.
Group 4: Can cause severe human disease.
Likely risk of spread into the community.
No effective prophylaxis and treatment.
The micro-organisms considered the most effective biological weapons are chosen because
they:
• are easily disseminated into the public or are highly infectious (readily transmitted
from person to person).
• cause high mortality.
• require special action for public health control
The organisms mostly fall into Hazard Group 4. Viruses such as Variola major virus
(smallpox) and Ebola and Marburg viruses are not easily treated with antiviral agents but
there are effective vaccines available (although not routinely used). Bacillus anthracis and Yersinia pestis(the aetiological agent of plague) can be treated with antibiotics.
Cited By Anil Bhujel
Bsc Microbiology,TU.
Microbiology Student at PBPC, Nayabazzar-9, Pokhara.
SOME SUGGESTED REFERENCES:
www.cdc.gov/flu/about/qa/antiviralresistance.htm
www.cdc.gov/flu/professionals/antivirals/antiviral-drug-resistance.htm
www.ncbi.nlm.nih.gov/pubmed/1706571
www.who.int/.../Penn_Points_to_consider_on_antiviral_drug_resistance....
www.ncbi.nlm.nih.gov/pubmed/18284179
www.psfk.com/2013/02/newspapers-epidemic-prediction-database.html
ucdavismagazine.ucdavis.edu/issues/su06/feature_1c.html
en.wikipedia.org/wiki/Pandemic
www.bbc.com/future/story/20130711-what-if-a-pandemic-strikes
www.homelandsecuritynewswire.com/topics/epidemics-pandemics
Bsc Microbiology,TU.
Microbiology Student at PBPC, Nayabazzar-9, Pokhara.
SOME SUGGESTED REFERENCES:
www.cdc.gov/flu/about/qa/antiviralresistance.htm
www.cdc.gov/flu/professionals/antivirals/antiviral-drug-resistance.htm
www.ncbi.nlm.nih.gov/pubmed/1706571
www.who.int/.../Penn_Points_to_consider_on_antiviral_drug_resistance....
www.ncbi.nlm.nih.gov/pubmed/18284179
www.psfk.com/2013/02/newspapers-epidemic-prediction-database.html
ucdavismagazine.ucdavis.edu/issues/su06/feature_1c.html
en.wikipedia.org/wiki/Pandemic
www.bbc.com/future/story/20130711-what-if-a-pandemic-strikes
www.homelandsecuritynewswire.com/topics/epidemics-pandemics
.






Comments