MICROBIAL INFECTIONS OF HUMANS(HUMAN MICROBIOLOGY CONTD..)
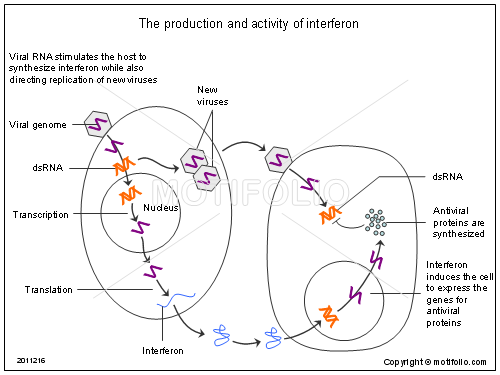
PRODUCTION OF INTERFERON:
It was demonstrated in the 1950s that the culture medium bathing virus infected cell lines could inhibit
the multiplication of viruses in separate flasks. The active substances, termed interferons, are cytokines
(as discussed above) and are produced within hours of virus infection to limit the spread of virus in the
host whilst specific immune responses are developing. Most cell types can synthesise interferon but they
can be grouped into three broad types according to the cell type that manufactures them.
• IFN- (alpha): leucocyte interferon,IFN- (beta): fibroblast interferon,
• IFN- (delta): immune interferon (activated T-lymphocytes and NK cells).
Double-stranded RNA is the most potent stimulus for the production and release of interferon alpha and
beta which act to prevent or limit the surrounding cells from becoming infected. Interferons are
prophylactic rather than curative. The actions of interferons are somewhat varied in that they can
modulate the activity of immune cells as well as induce resistance to viral infection. Two important
antiviral processes induced by interferon in virus-infected cells are:
• the inhibition of viral-induced protein synthesis, and
• degradation of viral mRNA and rRNA.
The central player in cells stimulated by IFN is protein kinase R(PKR). The letter R is taken from the
viral dsRNA that activates the transcription of this protein kinase. PKR produced by cells stimulated by
IFN inhibits viral driven protein synthesis by binding to the double-stranded RNA. PKR will also trigger
apoptosis, presumably as a last resort following uncontrolled viral replication.
TRIGGERING APOPTOSIS:
Controlled cell death (apoptosis) is a mechanism by which multicellular organisms remove unwanted
cells either during development (wonderfully illustrated by the loss of the tail in the developing tadpole) or respond to genomic abnormalities (pre-cancerous changes).
Apoptosis is also of great value as a defence mechanism against virus infection in cells. Apoptosis, in
contrast to necrosis, is a tightly controlled process that leaves no mess and causes no damage to
surrounding cells. As with host cell nucleases, viruses have acquired mechanisms with which to
counteract the triggering of apoptosis.
VIRUS-INDUCED TISSUE DAMAGE: VIRUS OR HOST?
To what extent does the replication of the virus in the tissue cause the disease? Some of these help explain why virus infections can cause cell damage. For example, lytic infections will result in the loss of function of the infected cells. If sufficient numbers of cells are damaged in this way then the function of the organ may
be compromised. Rotaviruses cause diarrhoea in humans and animals. The infected cells of the villi of
the ileum are shed resulting in the loss of absorptive surface area contributing to the diarrhoea. The
diarrhoea is a direct consequence of the viral damage in the enterocytes. Other examples of tissue
damage are indirect. In true clinical poliomyelitis the virus damages the nerve cells that serve the muscle
cells of the limbs. The damage to the nerve cells by the virus causes the muscle to atrophy. Virus cannot
be found in the affected muscles themselves. Furthermore, poliomyelitis virus normally only multiplies
within the enterocytes of the small intestine and they show no morphological alterations. This serves to
remind us that the CPE observed in the cell lines used to grow viruses in the laboratory are not
necessarily reflected in the natural host cells.
The common symptoms of many viral infections, myalgia and headache, are usually indirect
consequences of the host immune response (often cytokines such as tumour necrosis factors)
rather than direct viral replication in the muscles and brain.
Different infection strategies are employed by different viruses. The extremes are an acute, hit-and-run
approach or a chronic persistent type of infection. Viruses choosing the former option will have less
concern for the effects of rapid viral multiplication on the host. Such viruses will tend to suppress or
inactivate acute (innate) mechanisms of host defence so as to gain time for the rapid multiplication of
new virus. If the host is fatally damaged by the infection, as long as sufficient virus has been
manufactured such that the infection is transmitted to new hosts, then the death of the host is of no
concern. Aggressive virus multiplication will probably result in protective immunity such that, if the
host recovers, he or she will not be available for the virus to reinfect. Chronic infections will need to
adopt those strategies that deal with the longer term problems of specific immunity, i.e. the development
of specific antibodies and cell-mediated cytotoxicity. The reaction of the host to intracellular parasites, viruses in particular, will often result in tissue damage.
IMMUNE CELL-MEDIATED CYTOTOXICITY
In tissues with rapid turnover, virus-infected cells that are lost may be replaced (e.g. intestinal and skin
epithelia). In non-replicating tissues like the heart and nerve tissue virus-infected cells cannot be lost so
readily without possible functional deficit. If, therefore, the immune response damages these cells, the
organ suffers from impaired function. Hepatitis viruses do not cause lytic infections of the hepatocyte
but instead evoke a lymphocytic cell inflammation. This cell-mediated host response is what damages
the liver tissue with resulting impairment of function.
IMMUNE COMPLEX REACTIONS
Immune complex reactions are typical of persistent infections. The antigen–antibody complexes can get
stuck (‘deposit’ themselves) in arteries to cause vasculitis (which manifests itself as a skin rash) or in the basement membrane of the glomeruli to cause kidney damage. Both are seen with chronic hepatitis B infections.
Cited By Kamal Singh Khadka
Msc Microbiology, TU.
Assistant Professor in PU, PBPC, PNC, LA, NA.
Pokhara, Nepal.
SOME SUGGESTED REFERENCES:
en.wikipedia.org/wiki/Interferon
bloodjournal.hematologylibrary.org/content/109/3/1165.full.pdf
www.britannica.com/EBchecked/topic/290230/interferon
courses.bio.indiana.edu/M430-Taylor/interferon.html
faculty.smu.edu/jbuynak/Interferons%20power%20pt.ppt
www.ncbi.nlm.nih.gov › NCBI › Literature › Bookshelf
en.wikipedia.org/wiki/Apoptosis
www.promega.com › Resources › Multimedia
users.rcn.com/jkimball.ma.ultranet/BiologyPages/A/Apoptosis.html
www.biooncology.com/research-education/apoptosis/pathways
en.wikipedia.org/wiki/Antibody-dependent_cell-mediated_cytotoxicity
medical-dictionary.thefreedictionary.com/cell-mediated+cytotoxicity
immuneweb.xxmu.edu.cn/reading/81.pdf
www.medterms.com/script/main/art.asp?articlekey=10097
www.ncbi.nlm.nih.gov › NCBI › Literature › Bookshelf
www.umich.edu/~bmsteach/lopatin/.../05T-EffectorMechanisms03.ppt
It was demonstrated in the 1950s that the culture medium bathing virus infected cell lines could inhibit
the multiplication of viruses in separate flasks. The active substances, termed interferons, are cytokines
(as discussed above) and are produced within hours of virus infection to limit the spread of virus in the
host whilst specific immune responses are developing. Most cell types can synthesise interferon but they
can be grouped into three broad types according to the cell type that manufactures them.
• IFN- (alpha): leucocyte interferon,IFN- (beta): fibroblast interferon,
• IFN- (delta): immune interferon (activated T-lymphocytes and NK cells).
Double-stranded RNA is the most potent stimulus for the production and release of interferon alpha and
beta which act to prevent or limit the surrounding cells from becoming infected. Interferons are
prophylactic rather than curative. The actions of interferons are somewhat varied in that they can
modulate the activity of immune cells as well as induce resistance to viral infection. Two important
antiviral processes induced by interferon in virus-infected cells are:
• the inhibition of viral-induced protein synthesis, and
• degradation of viral mRNA and rRNA.
The central player in cells stimulated by IFN is protein kinase R(PKR). The letter R is taken from the
viral dsRNA that activates the transcription of this protein kinase. PKR produced by cells stimulated by
IFN inhibits viral driven protein synthesis by binding to the double-stranded RNA. PKR will also trigger
apoptosis, presumably as a last resort following uncontrolled viral replication.
TRIGGERING APOPTOSIS:
Controlled cell death (apoptosis) is a mechanism by which multicellular organisms remove unwanted
cells either during development (wonderfully illustrated by the loss of the tail in the developing tadpole) or respond to genomic abnormalities (pre-cancerous changes).
Apoptosis is also of great value as a defence mechanism against virus infection in cells. Apoptosis, in
contrast to necrosis, is a tightly controlled process that leaves no mess and causes no damage to
surrounding cells. As with host cell nucleases, viruses have acquired mechanisms with which to
counteract the triggering of apoptosis.
VIRUS-INDUCED TISSUE DAMAGE: VIRUS OR HOST?
To what extent does the replication of the virus in the tissue cause the disease? Some of these help explain why virus infections can cause cell damage. For example, lytic infections will result in the loss of function of the infected cells. If sufficient numbers of cells are damaged in this way then the function of the organ may
be compromised. Rotaviruses cause diarrhoea in humans and animals. The infected cells of the villi of
the ileum are shed resulting in the loss of absorptive surface area contributing to the diarrhoea. The
diarrhoea is a direct consequence of the viral damage in the enterocytes. Other examples of tissue
damage are indirect. In true clinical poliomyelitis the virus damages the nerve cells that serve the muscle
cells of the limbs. The damage to the nerve cells by the virus causes the muscle to atrophy. Virus cannot
be found in the affected muscles themselves. Furthermore, poliomyelitis virus normally only multiplies
within the enterocytes of the small intestine and they show no morphological alterations. This serves to
remind us that the CPE observed in the cell lines used to grow viruses in the laboratory are not
necessarily reflected in the natural host cells.
The common symptoms of many viral infections, myalgia and headache, are usually indirect
consequences of the host immune response (often cytokines such as tumour necrosis factors)
rather than direct viral replication in the muscles and brain.
Different infection strategies are employed by different viruses. The extremes are an acute, hit-and-run
approach or a chronic persistent type of infection. Viruses choosing the former option will have less
concern for the effects of rapid viral multiplication on the host. Such viruses will tend to suppress or
inactivate acute (innate) mechanisms of host defence so as to gain time for the rapid multiplication of
new virus. If the host is fatally damaged by the infection, as long as sufficient virus has been
manufactured such that the infection is transmitted to new hosts, then the death of the host is of no
concern. Aggressive virus multiplication will probably result in protective immunity such that, if the
host recovers, he or she will not be available for the virus to reinfect. Chronic infections will need to
adopt those strategies that deal with the longer term problems of specific immunity, i.e. the development
of specific antibodies and cell-mediated cytotoxicity. The reaction of the host to intracellular parasites, viruses in particular, will often result in tissue damage.
IMMUNE CELL-MEDIATED CYTOTOXICITY
In tissues with rapid turnover, virus-infected cells that are lost may be replaced (e.g. intestinal and skin
epithelia). In non-replicating tissues like the heart and nerve tissue virus-infected cells cannot be lost so
readily without possible functional deficit. If, therefore, the immune response damages these cells, the
organ suffers from impaired function. Hepatitis viruses do not cause lytic infections of the hepatocyte
but instead evoke a lymphocytic cell inflammation. This cell-mediated host response is what damages
the liver tissue with resulting impairment of function.
IMMUNE COMPLEX REACTIONS
Immune complex reactions are typical of persistent infections. The antigen–antibody complexes can get
stuck (‘deposit’ themselves) in arteries to cause vasculitis (which manifests itself as a skin rash) or in the basement membrane of the glomeruli to cause kidney damage. Both are seen with chronic hepatitis B infections.
Cited By Kamal Singh Khadka
Msc Microbiology, TU.
Assistant Professor in PU, PBPC, PNC, LA, NA.
Pokhara, Nepal.
SOME SUGGESTED REFERENCES:
en.wikipedia.org/wiki/Interferon
bloodjournal.hematologylibrary.org/content/109/3/1165.full.pdf
www.britannica.com/EBchecked/topic/290230/interferon
courses.bio.indiana.edu/M430-Taylor/interferon.html
faculty.smu.edu/jbuynak/Interferons%20power%20pt.ppt
www.ncbi.nlm.nih.gov › NCBI › Literature › Bookshelf
en.wikipedia.org/wiki/Apoptosis
www.promega.com › Resources › Multimedia
users.rcn.com/jkimball.ma.ultranet/BiologyPages/A/Apoptosis.html
www.biooncology.com/research-education/apoptosis/pathways
en.wikipedia.org/wiki/Antibody-dependent_cell-mediated_cytotoxicity
medical-dictionary.thefreedictionary.com/cell-mediated+cytotoxicity
immuneweb.xxmu.edu.cn/reading/81.pdf
www.medterms.com/script/main/art.asp?articlekey=10097
www.ncbi.nlm.nih.gov › NCBI › Literature › Bookshelf
www.umich.edu/~bmsteach/lopatin/.../05T-EffectorMechanisms03.ppt













Comments