MICROBIAL INFECTIONS OF HUMANS(HUMAN MICROBIOLOGY CONTD..)
INTRACELLULAR PARASITES:
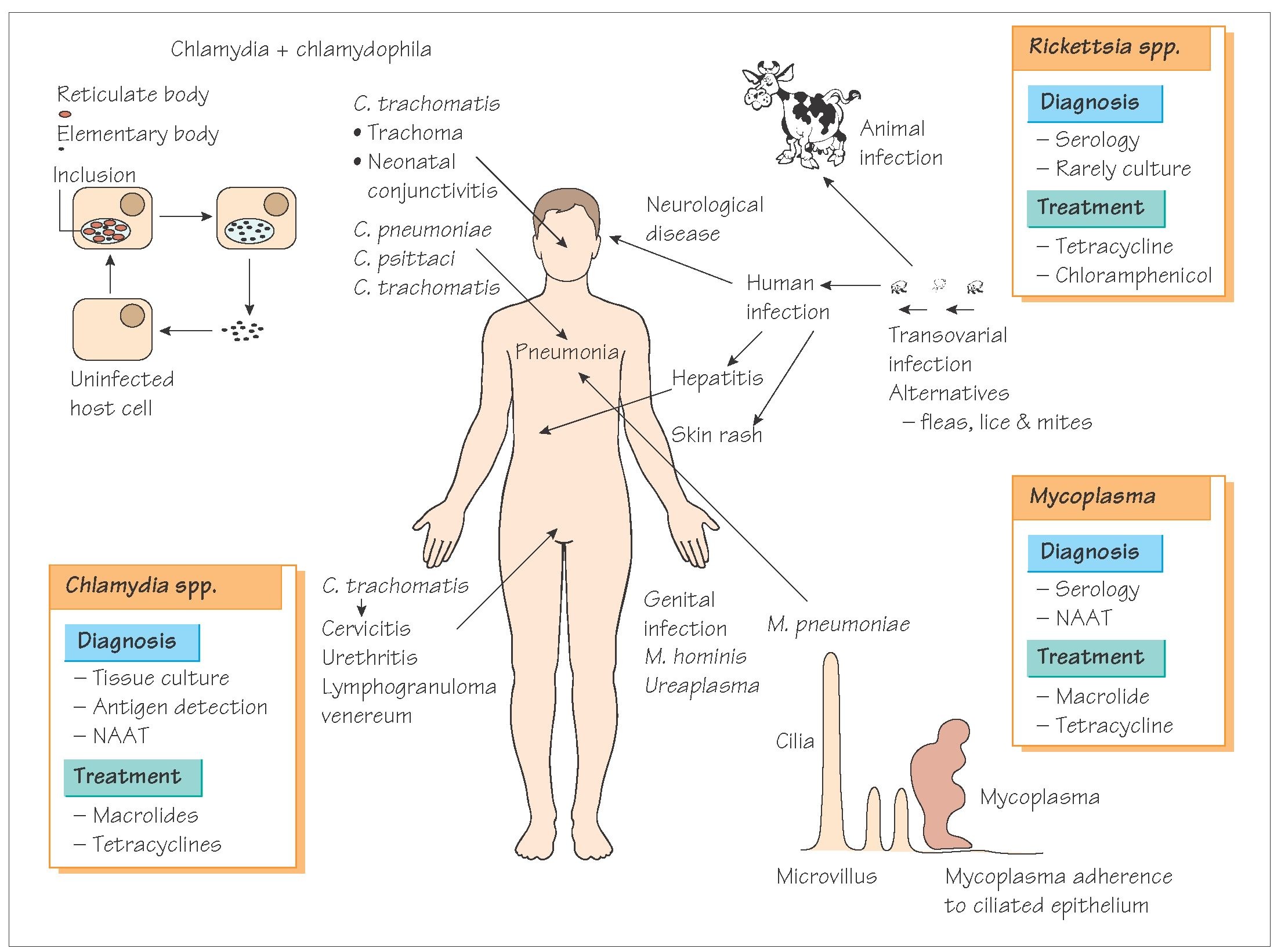
The genera Chlamydia and Rickettsia both contain species that cause human disease. Chlamydia
trachomatis causes trachoma and Rickettsia prowazekii causes typhus fever. Studies of the genomes of
these intracellular bacteria have yielded interesting patterns. By utilising host nutrients, these organisms
have lost the corresponding biosynthetic pathways; the amino acid synthesis pathways are absent with
the organisms choosing to utilise those of the host cell. Similarly the enzymes for synthesis of purines
and pyrimidines are largely absent. Energy requirements of these bacteria are still paramount but they
have lost the need for anaerobic glycolytic pathways as they now live in an aerobic environment. The
reduction in requirement for such enzymes results in the reduction in size of the genome of the bacteria.
The striking feature of these bacteria is the small genome size, around 1 megabase (1 Mb), roughly
equivalent to 800–900 genes, compared with free-living bacteria such as Esch. coli with a genome of 4.6
Mb in size. There are, however, pressures. The genomes of these intracellular parasites exhibit mutation
rates higher than free-living bacteria. One of the reasons is the loss of DNA-mismatch repair enzymes.
Another reason is the reduced opportunities for recombination or horizontal gene transfer with other
bacteria when choosing an obligate intracellular replication strategy. A similar problem is seen with
viruses. The intracellular existence will be dominated by the considerably larger host genome so it is
economical to lose any duplicated bacterial genes. Finally, the asexual, clonal replication with this
reduced genome repair kit will result in the accumulation of neutral and deleterious mutations, which
cannot be removed by selective pressure. Such a scenario might suggest that the organism has not got
much hope of survival. However, the organism can devote a dominant proportion of its genome to code
for features that favour persistence in the face of host defences.
ENDOSYMBIONTS:
As with obligate intracellular parasites, the asexual, clonal strategy results in a reduced genome with a
high proportion of mutated code. The residual genome codes for essential cell functions such as growth
and division. In contrast to obligate intracellular parasites, the host benefits from housing endosymbionts
such as Buchnera spp.because they synthesise amino acids that are available to the host as well. This
contrasts with Chlamydia and Rickettsia, both of which have lost the amino acid biosynthetic capability
and therefore compete for amino acids with the host. The gene loss has consequences for other cell
functions. An endosymbiont has no need for genes that code for antigenic variation and other defensive
host protection mechanisms. Likewise the organism has no need for mechanisms of entering and
leaving the host cell as it has become a stable partner. The conflict of interest between the organism and the host will result in a reciprocal exploitation that may result in mutualism or parasitism. One of the advantages of becoming an endosymbiont is efficient transmission from host to offspring (vertical transmission) and this benefits the host as well as the symbiont, whereas the genetic isolation and increased mutation rate might be seen as a disadvantage. The accumulation of deleterious mutations in a genetically isolated population (with almost no opportunities for recombination) is known as Muller’s ratchet. Endosymbionts will suffer from this
gradual loss of fitness through Muller’s ratchet. Conversely, the obligate intracellular parasite is usually
spread between unrelated hosts (horizontal transmission) and increases the chances of recombination,
and thus conflict between host and bacterium.
The genera Chlamydia and Rickettsia both contain species that cause human disease. Chlamydia
trachomatis causes trachoma and Rickettsia prowazekii causes typhus fever. Studies of the genomes of
these intracellular bacteria have yielded interesting patterns. By utilising host nutrients, these organisms
have lost the corresponding biosynthetic pathways; the amino acid synthesis pathways are absent with
the organisms choosing to utilise those of the host cell. Similarly the enzymes for synthesis of purines
and pyrimidines are largely absent. Energy requirements of these bacteria are still paramount but they
have lost the need for anaerobic glycolytic pathways as they now live in an aerobic environment. The
reduction in requirement for such enzymes results in the reduction in size of the genome of the bacteria.
The striking feature of these bacteria is the small genome size, around 1 megabase (1 Mb), roughly
equivalent to 800–900 genes, compared with free-living bacteria such as Esch. coli with a genome of 4.6
Mb in size. There are, however, pressures. The genomes of these intracellular parasites exhibit mutation
rates higher than free-living bacteria. One of the reasons is the loss of DNA-mismatch repair enzymes.
Another reason is the reduced opportunities for recombination or horizontal gene transfer with other
bacteria when choosing an obligate intracellular replication strategy. A similar problem is seen with
viruses. The intracellular existence will be dominated by the considerably larger host genome so it is
economical to lose any duplicated bacterial genes. Finally, the asexual, clonal replication with this
reduced genome repair kit will result in the accumulation of neutral and deleterious mutations, which
cannot be removed by selective pressure. Such a scenario might suggest that the organism has not got
much hope of survival. However, the organism can devote a dominant proportion of its genome to code
for features that favour persistence in the face of host defences.
ENDOSYMBIONTS:
As with obligate intracellular parasites, the asexual, clonal strategy results in a reduced genome with a
high proportion of mutated code. The residual genome codes for essential cell functions such as growth
and division. In contrast to obligate intracellular parasites, the host benefits from housing endosymbionts
such as Buchnera spp.because they synthesise amino acids that are available to the host as well. This
contrasts with Chlamydia and Rickettsia, both of which have lost the amino acid biosynthetic capability
and therefore compete for amino acids with the host. The gene loss has consequences for other cell
functions. An endosymbiont has no need for genes that code for antigenic variation and other defensive
host protection mechanisms. Likewise the organism has no need for mechanisms of entering and
leaving the host cell as it has become a stable partner. The conflict of interest between the organism and the host will result in a reciprocal exploitation that may result in mutualism or parasitism. One of the advantages of becoming an endosymbiont is efficient transmission from host to offspring (vertical transmission) and this benefits the host as well as the symbiont, whereas the genetic isolation and increased mutation rate might be seen as a disadvantage. The accumulation of deleterious mutations in a genetically isolated population (with almost no opportunities for recombination) is known as Muller’s ratchet. Endosymbionts will suffer from this
gradual loss of fitness through Muller’s ratchet. Conversely, the obligate intracellular parasite is usually
spread between unrelated hosts (horizontal transmission) and increases the chances of recombination,
and thus conflict between host and bacterium.
THE TERMS:
From here on we will concentrate on microbial associations with humans, especially those that are
detrimental to health. It is necessary to provide some further definitions of the common terms.
PARASITE:
The parasite can be qualified further as an ectoparasite, where it exists on the outer surfaces of the host,
in contrast to an endoparasite, one that is found inside the host. Multicellular parasites such as flukes
and worms are usually referred to as macroparasites and can be considered separately from the
microparasites we have introduced above (viruses, bacteria). Macroparasites have adapted some of
their structural features such that they have either disappeared or reduced in size, e.g. organs involved in
movement, organs for storing food, complicated nervous systems, lengthy intestinal tract (in fact,
tapeworms have no intestinal tract). With bacteria there are no such structural differences between strict
parasitic species and harmless species.
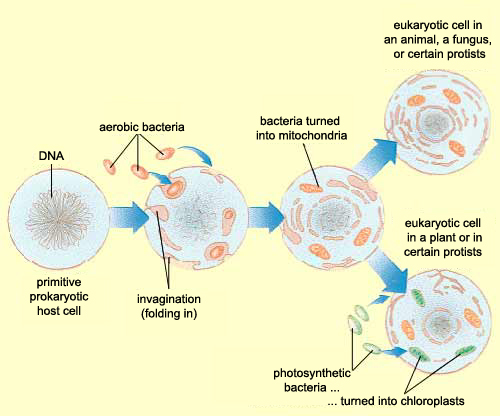
ORIGINS OF EUKARYOTIC INTRACELLULAR ORGANELLES
Mitochondria appear to be bacteria that have undergone such drastic genome reduction that they are
now obligate intracellular partners in the host; in fact, genes are donated to the host genome.
Whereas endosymbionts and intracellular parasites infect only certain cells, mitochondria are present
in virtually all cells of the body. Mitochondria still provide benefits for the host cell: they are net
exporters of energy but cannot replicate independently of the host cell.
PATHOGEN:
This is a micro-organism that can cause disease. All organisms can cause disease under appropriate
circumstances but for some micro-organisms this is much more likely. These organisms are called
‘primary pathogens’, whereas those that need special circumstances are called ‘secondary pathogens’. It
might seem unnecessary to distinguish between parasite and pathogen but they are not synonymous. An
organism may be one but not the other. Whilst it is not difficult to imagine that a parasite may cause no
overt (clinically apparent) disease, i.e., is not pathogenic, it is possible that organisms are pathogenic but
not parasitic. Consider food poisoning caused by Staphylococcus aureus and Clostridium botulinum.
The toxins are released by the organisms when they multiply in the food. Thus humans ingest preformed toxin and develop symptoms of vomiting and botulism respectively. In this instance both
organisms are pathogenic but not parasitic to man.
INFECTION:
Someone is infected if the microbe has successfully colonised or at least established itself, albeit
temporarily, on a host. If they develop symptoms of disease they have a clinical infection. Alternatively,
if the infection does not result in symptoms the person is infected but asymptomatic.
COLONISATION:
This describes the presence of micro-organisms, pathogenic or non-pathogenic, in large numbers, on a
host without tissue damage or clinical disease.
DISEASE
This is a condition of disordered well-being. In terms of infection, this means the pathogen is able to
cause pathological damage, that is, tissue damage.
INFECTIOUS DISEASE
Also referred to as a communicable disease, an infectious disease is an infection that can be transmitted
between humans (or organisms/animals, etc.). The organism is able to spread from one host to another
and is therefore infectious. The term contagious means the same but is less frequently used. Not all infections are infectious, i.e. are not naturally transmissible from one person to another (e.g. brain
abscess, osteomyelitis, infective endocarditis).
A LITTLE MORE ON PARASITISM:
The above terms can be viewed/amended in terms of humans and micro-organisms. Rather than a series of distinct definitions, the above terms represent a spectrum of association. Where humans represent the host environment the relationship bacteria found on humans, they are able to multiply outside
of the host when suitable nutrient between the host and the normal microbial flora is mostly mutualistic, but under certain circumstances when members of the normal flora are able to cause disease, the relationship will be parasitic, and often harmful. With viruses, it is clear that they are obligate intracellular parasites. They utilise the host synthetic machinery to reproduce. The association between virus and host cell is absolute and the virus will only replicate at the expense of the host. The extent of the association between parasite and host may be determined by the biochemical dependence of the parasite on the host. For bacs are provided.
With bacteria parasitic to humans, the organisms have evolved such that they obtain essential nutrients
directly from the host rather than from the environment or de novosynthesis. There are indications that
organisms with a restricted metabolic capability (for example, within the genus Salmonella) are those
that cause the more severe infections. This may also account for species specificity as exemplified by
Salmonella dublin which infects cattle whereas Salmonella choleraesuis infects pigs.
SOME SUGGESTED REFERENCES:
Cited By Kamal Singh Khadka
Msc Microbiology, TU.
Assistant Professor In Pokhara University, Pokhara Bigyan Tatha Prabidhi Campus, PNC, LA, NA.
Pokhara, Nepal.




Comments