MICROBIAL BIOTECHNOLOGY: SCOPE, TECHNIQUES CONTD
ENVIRONMENTAL APPLICATIONS OF MICROORGANISMS:
Microorganisms mitigate a multitude of impacts that result from human
use of the natural resources of the planet. First and foremost, the essential
role of microorganisms in the treatment of waste water is critical to the well being of life on Earth. Bio-remediation, bio-mining, and microbial desulfurization of coal are other large-scale processes in which important positive environmental outcomes are achieved by directly exploiting the combined metabolic capabilities of naturally occurring communities of microorganisms. In such applications, the functioning of a particular microbial community can be influenced through the manipulation of conditions (e.g., nutrients, oxygen tension, temperature, agitation).
1) WASTE WATER TREATMENT:
Living organisms consist of about 70% water. A human being, for instance,
has to consume an average of 1.5 L/day to survive.Freshwater represents only
about 2.5% of the water on the planet and is now a scarce resource
in many parts of the world. The volume of water being contaminated and
the need to reclaim waste water are increasing with the growth in population and industrial use. Waste water originates from four primary sources: sewage,industrial effluents, agricultural runoff, and storm water and urban runoff. Treatment of waste water is essential to prevent contamination of drinking water and entry of pathogens and contaminants in food chain.
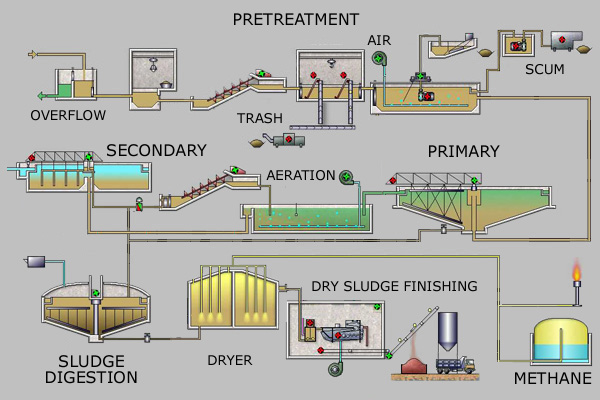
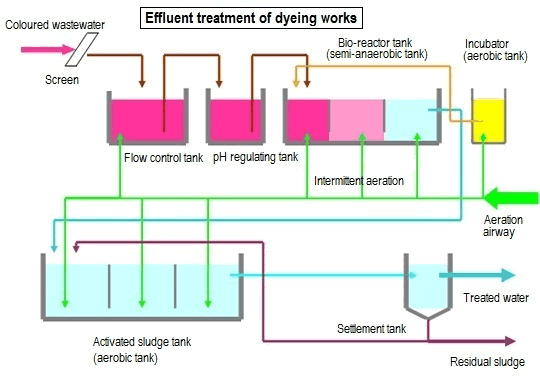
Primary treatment of sewage consists of removal of suspended solids. The secondary treatment of
sewage reduces the biochemical oxygen demand. This is accomplished by lowering the organic compound content of the effluent from the primary treatment through microbial oxidation by an incompletely characterized community of microorganisms in “activated sludge.” Bacteria of Zoogloea species play an important role in the aerobic secondary stage of sewage treatment. These organisms produce abundant extra-cellular polysaccharide and, as a result, form aggregates called flocs. Such aggregates efficiently adsorb organic matter, part of which is then metabolized by the bacteria.
The flocs settle out and are transferred to an anaerobic digester, where other bacteria complete the degradation of the adsorbed organic matter.
The microbial communities in a water treatment plant convert organic carbon to carbon dioxide, water, and sludge; convert some 80%of the ammonia and nitrate to molecular nitrogen; remove some soluble phosphate
through incorporation into the sludge, either as polyphosphate granules within bacterial cells or as struvite (crystalline MgNH4PO4); and remove pathogenic bacteria.
However, serious challenges in waste water treatment have yet to be fully addressed. The level of residual fixed nitrogen compounds and of phosphate in the effluent is still high enough to pose risks of eutrophication in the receiving bodies of water. Residues of many widely used pharmaceuticals present in municipal waste-water are incompletely removed and emerge in the effluent. Some of these compounds are biologically active at nano grams per liter and have demonstrable undesirable environmental effects. The chemical industry uses thousands of synthetic organic compounds in huge amounts,and many of these (or their degradation products) pose a similar concern to the pharmaceutical ones. Finally, waste-water treatment consumes energy but converts much of the ammonia and nitrate to nitrogen gas, and a significant amount of the phosphate remains in the effluent. Alternative processes that would recover the fixed nitrogen compounds and phosphate would offset the energy and economic costs of manufacturing the corresponding amount of chemical fertilizer and lessen the acute environmental problems associated with elevated nutrient levels in aquatic ecosystems.
NOTE: Biological Oxygen Demand
Maintenance of high oxygen concentration in aquatic ecosystems is essential for
the survival of fish and other aquatic organisms. Decomposition of organic matter
may rapidly deplete the oxygen. When organic matter such as untreated sewage
is added to an aquatic ecosystem, it is degraded by bacteria that consume oxygen
in the process. The biological oxygen demand (BOD) is related to the amount of
organic matter in the water. Usually, the oxygen consumption is measured over
a period of five days and is abbreviated BOD5. BOD5 for municipal wastewater
generally ranges from 80 to 250 mg O2 per liter. Appropriate secondary treatment
decreases the BOD5 to less than 20 mg O2 per liter.
Cited By Kamal Singh Khadka/ Shailendra Parajuli
Msc Microbiology, TU.
Assistant Professor In Pokhara University, PBTPC, PNC, LA, NA.
Pokhara, Nepal.
SOME SUGGESTED REFERENCES :
www.highveld.com/microbiology/environmental-microbiology.htm
textbookofbacteriology.net/Impact.html
www.lbl.gov/Science-Articles/Archive/microorganisms-phylotypes.html
www.researchgate.net/...microorganisms...environmental/.../9fcfd50abeb.
www.ncbi.nlm.nih.gov/pubmed/20662374
www.academia.edu/.../Role_of_Microorganisms_on_Wastewater_Treatm.
bartec-benke.nl/media/.../thebiologicalbasisofwastewatertreatment.pdf
kyocp.wordpress.com/.../types-of-bacteria-used-in-wastewater-treatment/
www.microtack.com/html/natural_sewage_treatment.htm
www.riparia.org.rs/aqua/activated-sludge.htm
mimoza.marmara.edu.tr/~neslihan.semerci/ENVE100/ENVE100Ch5.pdf
ic.ucsc.edu/~saltikov/bio119/lecture/25_lecture_wastewater_part.pdf
https://www.boundless.com/.../wastewater-treatment.../wastewater-and-se..
binodpandey.files.wordpress.com/2011/02/waste-water.pdf
https://www.theseus.fi/bitstream/handle/10024/.../Regmi_Shakil.pdf?...1
www.nepjol.info › Home › Our Nature › Vol 6, No 1 (2008) › Rajbanshi
www.ncbi.nlm.nih.gov › NCBI › Literature › PubMed Central (PMC)
.
Microorganisms mitigate a multitude of impacts that result from human
use of the natural resources of the planet. First and foremost, the essential
role of microorganisms in the treatment of waste water is critical to the well being of life on Earth. Bio-remediation, bio-mining, and microbial desulfurization of coal are other large-scale processes in which important positive environmental outcomes are achieved by directly exploiting the combined metabolic capabilities of naturally occurring communities of microorganisms. In such applications, the functioning of a particular microbial community can be influenced through the manipulation of conditions (e.g., nutrients, oxygen tension, temperature, agitation).
1) WASTE WATER TREATMENT:
Living organisms consist of about 70% water. A human being, for instance,
has to consume an average of 1.5 L/day to survive.Freshwater represents only
about 2.5% of the water on the planet and is now a scarce resource
in many parts of the world. The volume of water being contaminated and
the need to reclaim waste water are increasing with the growth in population and industrial use. Waste water originates from four primary sources: sewage,industrial effluents, agricultural runoff, and storm water and urban runoff. Treatment of waste water is essential to prevent contamination of drinking water and entry of pathogens and contaminants in food chain.
Primary treatment of sewage consists of removal of suspended solids. The secondary treatment of
sewage reduces the biochemical oxygen demand. This is accomplished by lowering the organic compound content of the effluent from the primary treatment through microbial oxidation by an incompletely characterized community of microorganisms in “activated sludge.” Bacteria of Zoogloea species play an important role in the aerobic secondary stage of sewage treatment. These organisms produce abundant extra-cellular polysaccharide and, as a result, form aggregates called flocs. Such aggregates efficiently adsorb organic matter, part of which is then metabolized by the bacteria.
The flocs settle out and are transferred to an anaerobic digester, where other bacteria complete the degradation of the adsorbed organic matter.
The microbial communities in a water treatment plant convert organic carbon to carbon dioxide, water, and sludge; convert some 80%of the ammonia and nitrate to molecular nitrogen; remove some soluble phosphate
through incorporation into the sludge, either as polyphosphate granules within bacterial cells or as struvite (crystalline MgNH4PO4); and remove pathogenic bacteria.
However, serious challenges in waste water treatment have yet to be fully addressed. The level of residual fixed nitrogen compounds and of phosphate in the effluent is still high enough to pose risks of eutrophication in the receiving bodies of water. Residues of many widely used pharmaceuticals present in municipal waste-water are incompletely removed and emerge in the effluent. Some of these compounds are biologically active at nano grams per liter and have demonstrable undesirable environmental effects. The chemical industry uses thousands of synthetic organic compounds in huge amounts,and many of these (or their degradation products) pose a similar concern to the pharmaceutical ones. Finally, waste-water treatment consumes energy but converts much of the ammonia and nitrate to nitrogen gas, and a significant amount of the phosphate remains in the effluent. Alternative processes that would recover the fixed nitrogen compounds and phosphate would offset the energy and economic costs of manufacturing the corresponding amount of chemical fertilizer and lessen the acute environmental problems associated with elevated nutrient levels in aquatic ecosystems.
NOTE: Biological Oxygen Demand
Maintenance of high oxygen concentration in aquatic ecosystems is essential for
the survival of fish and other aquatic organisms. Decomposition of organic matter
may rapidly deplete the oxygen. When organic matter such as untreated sewage
is added to an aquatic ecosystem, it is degraded by bacteria that consume oxygen
in the process. The biological oxygen demand (BOD) is related to the amount of
organic matter in the water. Usually, the oxygen consumption is measured over
a period of five days and is abbreviated BOD5. BOD5 for municipal wastewater
generally ranges from 80 to 250 mg O2 per liter. Appropriate secondary treatment
decreases the BOD5 to less than 20 mg O2 per liter.
Cited By Kamal Singh Khadka/ Shailendra Parajuli
Msc Microbiology, TU.
Assistant Professor In Pokhara University, PBTPC, PNC, LA, NA.
Pokhara, Nepal.
SOME SUGGESTED REFERENCES :
www.highveld.com/microbiology/environmental-microbiology.htm
textbookofbacteriology.net/Impact.html
www.lbl.gov/Science-Articles/Archive/microorganisms-phylotypes.html
www.researchgate.net/...microorganisms...environmental/.../9fcfd50abeb.
www.ncbi.nlm.nih.gov/pubmed/20662374
www.academia.edu/.../Role_of_Microorganisms_on_Wastewater_Treatm.
bartec-benke.nl/media/.../thebiologicalbasisofwastewatertreatment.pdf
kyocp.wordpress.com/.../types-of-bacteria-used-in-wastewater-treatment/
www.microtack.com/html/natural_sewage_treatment.htm
www.riparia.org.rs/aqua/activated-sludge.htm
mimoza.marmara.edu.tr/~neslihan.semerci/ENVE100/ENVE100Ch5.pdf
ic.ucsc.edu/~saltikov/bio119/lecture/25_lecture_wastewater_part.pdf
https://www.boundless.com/.../wastewater-treatment.../wastewater-and-se..
binodpandey.files.wordpress.com/2011/02/waste-water.pdf
https://www.theseus.fi/bitstream/handle/10024/.../Regmi_Shakil.pdf?...1
www.nepjol.info › Home › Our Nature › Vol 6, No 1 (2008) › Rajbanshi
www.ncbi.nlm.nih.gov › NCBI › Literature › PubMed Central (PMC)
.jpg)
.jpg)
.jpg)


Comments