CLINICAL MICROBIOLOGY
9) Nitrate Reduction Test:
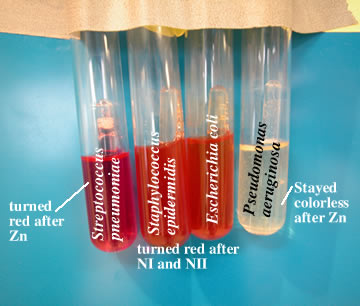
Many microorganisms can reduce nitrate salts such as potassium nitrate to nitrite salt, KNO2, under anaerobic conduction. This is an example of anaerobic respiration in which the nitrate molecule acts as electron acceptor in place of oxygen. This reduction of nitrate can easily be demonstrated experimentally by formulating a medium containing broth to which a very small amount of potassium nitrate is added.
Some mos possess the enzymatic capacity to act further on nitrates to reduce them to ammonia or molecule Nitrogen. After growing the organism in nitrate medium, its ability to reduce nitrate to nitrite is determined by the addition of two reagents solution A containing sulfanilic acid & solution B containing alpha-naphthylamine. Nitrite gives cherry red colored complex with sulfanilic acid & alpha-naphthylamine.
The culture may not produce red color due to : a) nitrates not reduced by mos or
b) the mos possess potent nitrate reductase which rapidly reduced nitrate beyond nitrites to ammonia or even molecular Nitrogen.
To determine these conditions,a small amount of Zn powder is added to the culture Zn reduces nitrates to nitrites. The development of red color on addition of Zn indicates that nitrates was not reduced by the organisms which suggests that organism is incapable of reducing nitrate to nitrite while if there is no color change after the addition of Zn to the culture suggests that nitrate is further reduced beyond nitrite to ammonia or even nitrogen.
Medical Application:
Most enteric bacteria are nitrate reducers. Pathogenic examples
include Escherichia coli (opportunistic urinary tract
infections), Klebsiella pneumoniae (bacterial pneumonia),
Morganella morganii and Proteus mirabilis (nosocomial
infections). Nonenteric nitrogen reducing pathogens include
Staphylococcus aureus (staphylococcal food poisoning,
bacteremia, various abscesses) and Bacillus anthracis
(anthrax).
Please visit links for references:
en.wikipedia.org/wiki/Nitrate_reductase_test
www.austincc.edu/microbugz/nitrate_reduction.php
www.vumicro.com › Home › Reference Books › Biochemical Tests
www.microbelibrary.org/...test/3660-nitrate-and-nitrite-reduction-test-pr...
www.sigmaaldrich.com/etc/medialib/docs/Fluka/.../73426dat.pdf
Cited By Kamal Singh Khadka
Assistant Professor in Pokhara University, Regional College Of Science & Technology,PNC,LA, NA
Pokhara, Nepal
Many microorganisms can reduce nitrate salts such as potassium nitrate to nitrite salt, KNO2, under anaerobic conduction. This is an example of anaerobic respiration in which the nitrate molecule acts as electron acceptor in place of oxygen. This reduction of nitrate can easily be demonstrated experimentally by formulating a medium containing broth to which a very small amount of potassium nitrate is added.
Some mos possess the enzymatic capacity to act further on nitrates to reduce them to ammonia or molecule Nitrogen. After growing the organism in nitrate medium, its ability to reduce nitrate to nitrite is determined by the addition of two reagents solution A containing sulfanilic acid & solution B containing alpha-naphthylamine. Nitrite gives cherry red colored complex with sulfanilic acid & alpha-naphthylamine.
The culture may not produce red color due to : a) nitrates not reduced by mos or
b) the mos possess potent nitrate reductase which rapidly reduced nitrate beyond nitrites to ammonia or even molecular Nitrogen.
To determine these conditions,a small amount of Zn powder is added to the culture Zn reduces nitrates to nitrites. The development of red color on addition of Zn indicates that nitrates was not reduced by the organisms which suggests that organism is incapable of reducing nitrate to nitrite while if there is no color change after the addition of Zn to the culture suggests that nitrate is further reduced beyond nitrite to ammonia or even nitrogen.
Medical Application:
Most enteric bacteria are nitrate reducers. Pathogenic examples
include Escherichia coli (opportunistic urinary tract
infections), Klebsiella pneumoniae (bacterial pneumonia),
Morganella morganii and Proteus mirabilis (nosocomial
infections). Nonenteric nitrogen reducing pathogens include
Staphylococcus aureus (staphylococcal food poisoning,
bacteremia, various abscesses) and Bacillus anthracis
(anthrax).
Please visit links for references:
en.wikipedia.org/wiki/Nitrate_reductase_test
www.austincc.edu/microbugz/nitrate_reduction.php
www.vumicro.com › Home › Reference Books › Biochemical Tests
www.microbelibrary.org/...test/3660-nitrate-and-nitrite-reduction-test-pr...
www.sigmaaldrich.com/etc/medialib/docs/Fluka/.../73426dat.pdf
web.clark.edu/rrausch/biolabs/260/NitrateReduction.pdf
www.mesacc.edu/~johnson/labtools/Dbiochem/nit.html
Cited By Kamal Singh Khadka
Assistant Professor in Pokhara University, Regional College Of Science & Technology,PNC,LA, NA
Pokhara, Nepal


Comments